 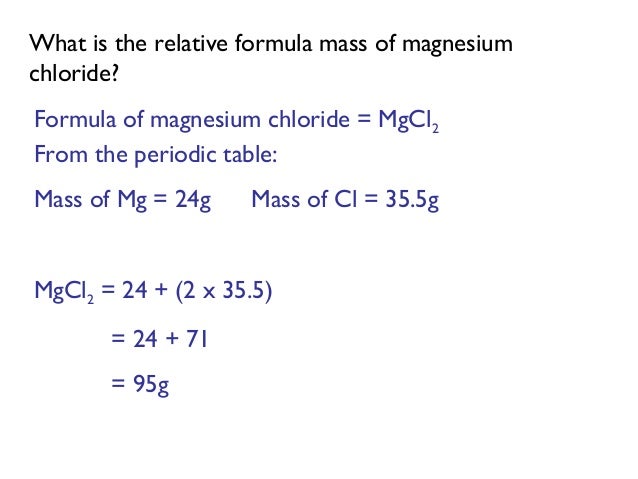
Even the cell membranes are made of proteins. Carbon is an important element for all living organisms, as it is used to construct the basic building blocks of life, such as carbohydrates, lipids, and nucleic acids. These elements form the basic building blocks of the major macromolecules of life, including carbohydrates, lipids, nucleic acids and proteins. The first four of these are the most important, as they are used to construct the molecules that are necessary to make up living cells. They are carbon, hydrogen, nitrogen, oxygen, phosphorus, and sulfur. In biology, the elements of life are the essential building blocks that make up living things. This one, I hope this whole concept is clear to you. 
So therefore the osmolarity of mg cl two will be three multiply by. So hence the number of particles are, are there three multiplied by and the modularity it is given here 0.3 M. So therefore the osmolarity or small clarity is equal to number of particles formed multiplied by polarity. So one particle of magnesium iron and two particles of chloride ions are there. It is so therefore there are three particles are here. Then the number of particles are three, so MG cl two, that means it gets dissociated into MG plus two plus two Cl -1. When MG cl two, it is dissolved in water. Further are small clarity, a similarity or 0.3 mm gcl to see the number of particles found by dissolving MG cl two in water. So this much of powder will make 200 ml of 0.5 M solution. So we can say that the mass of all the weight of MG cl two, it is 9.5 to 1 graham eaters. Therefore the weight of mg cl two Ingraham is equal to the number of moles multiply by moller, yep moral molecular wait, so number of moles are 0.14 MG cl two multiply by and and gcl two molecular weight is 95.21 and it comes to 95 9 point 5- one ground. So but we need to calculate the weight of this MG cl. Just write it as we're divided by molecular wait fine. Since we know we know that the number of moles is equal to the mass or the weight of the salute divided by the molecular weight or the molar mass. Now let us calculate the weight of mg cl two. 
It will contain 0.5 multiply by 0.2 moles of mg cl two that is 0.1 moles of mg cl two, magnesium chloride. So that means we are saying that one liter of solution it contains 0.5 moles of mg cl two. It has 0.5 mol of M d C LW right in in 200 ml. Therefore there will be in 0.5 M of mg cl two in the solution of mg cl do. Yes, so that means we can see it has one more all of mg cl two. It has one mole of magnesium chloride in one liter of solution. So we know that one M solution of magnesium chloride. No, we know that latest force calculate the weight of the magnesium chloride. So in this question we need to find out the weight of them, MG cl two that is magnesium chloride and also the osmolarity. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
